C2h4 Lewis Structure
Use information from step 4 and 5 to draw the lewis structure. There are only single bond between carbon atom and hydrogen atom because hydrogen caannot keep more than two electrons in its last shell.

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
Remember that hydrogen atoms always go on the outside of a Lewis structure and that they only need two valence.

C2h4 lewis structure. Drawing the Lewis structure for C 2 H 4 named ethene requires the use of a double bond. In a double bond two pairs of valence electrons are shared for a total of four valence electrons. In C2H4 if we look into the lewis structure we will see that there are three bonded pairs of electrons around each carbon and zero lone pair.
The solution is to share three pairs of valence electrons and. A lot can be studied about the molecule through the Lewis structure which says acetylene C2H2 is an unsaturated compound making it compatible and reactive enough to bond with atmospheric molecules and become toxic to human health. Carbon is in group 4 sometimes written 14 so it has 4 valence electrons.
Its slightly soluble in water. Metallic elements to the left of the staircase dividing line tend to methane ch4 is the classic example of this guideline in action. Start by forming covalent bonds between the Carbon and Hydrogen atoms.
If we come way over here to Hydrogen its in group 1. Lets take a look. To do that we always count our valence electrons up first.
Placing the Elements in the Drawing. No lone pair is present on the central or outer atom in the lewis structure of ethene. Its C2H4 and we want to write the dot structures for ethene.
Alternatively a dot method can be used to draw the lewis structure. Find the Total Number of Valence Electrons refer to the Instructions Below the Pictures Step 2. The ethylene is a covalent bond and lewis base.
C2H4 Lewis structure contains four C-H bonds and one double bond in between two carbon atoms. We place two valence electrons between each atom as shown in the figure. C2H4 Lewis Structure.
-169 C OU Chemical Safety Data No longer updated More details-169 C Jean-Claude Bradley Open Melting Point Dataset 15806 21322-1692 C Jean-Claude Bradley Open Melting Point Dataset 28095 28096-16935 C Jean-Claude Bradley Open Melting Point Dataset 28095 28096-169 C SynQuest 51680 1300-1-01-169 C Sigma-Aldrich SIAL-00489-1692 C Kaye Laby No longer updated. Count total valence electron in C2H4. It has 1 valence electron.
Lewis dot structure of C 2 H 4. Drawing the Lewis Structure for C 2 H 4. Therefore there cannot be more than one stable resonance structure for C 2 H 4.
Arrangement of atoms shown below dashed lines show connections between atoms. What are the steps to draw Lewis structures. For C 2 H 2 you have a total of 10 valence electrons to work with.
To draw the c2h4 lewis. Use the Rest of the Dots to Finish Your Lewis Structure. So I hope this video helps you to understand this and for more such videos on lowest structure molecular geometry Polarity of the molecules.
1 point for the correct selections assessed when you answer and 5 points for the Lewis structure on your work assessed when I review. Calculate the total valence electrons in the molecule. So this is the lowest structure of C two H four in which there is a double bond between two carbon atoms and the carbon atoms are forming single bonds with two hydrogen atoms over here.
However in Hydrocarbons we always place the Carbon atoms in the center as shown in the figure. Draw Two Electrons Between Atoms to Form the Chemical Bonds. Draw the Lewis structure for C2H4.
For C 2 H 4 you have a total of 12 total valence electrons. CO Lewis Structure In CO Lewis structurethe carbon atom follows the octet rule and the oxygen atom also follows the octet ruleSoCO follows the octet rule for all the atoms. According to the VSEPR chart the shape of the ethene molecule is trigonal planar.
We have 12 available valence electrons. Hydrogen is the least electronegative element here. In CO Lewis structurewe get five pairs of electronsOut of five pairs of electronsCO has three bond pairs and two lone pairs of electronsHere carbon has one lone pair and oxygen has one lone pair of electrons.
There are two triangles overlapping each other as we can see in the diagram. H-----C---H SHOW WORK This question is worth a total of 6 points. The lewis structure of C2H4 is very easy to draw-Some steps need to follow for drawing the C2H4 Lewis dot structure 1.
In drawing the Lewis structure for C 2 H 2 also called ethyne youll find that you dont have enough valence electrons available to satisfy the octet for each element if you use only single bonds. Drawing the Lewis Structure for C 2 H 2 Ethyne or Acetylene. Acetylene C2H2 is a toxic molecule for human beings as it can reduce the concentration of oxygen in the air.
I looked at the lewis structures they look similar but why is one opposite. Ethylene C2H4 has sp2 hybridization and its bond angle is between 1166 to 1217. In the lewis structure of C 2 H 4 there are only four C-H bonds one CC bond and no lone pairs on last shells.
The molecular geometry of C2H4 is trigonal planar above are the explanation of its Lewis structure. Based on vsepr allows us to determine if the entire molecule is polar or nonpolar.
![]()
Draw And Explain The Lewis Structure Of C2h4 Study Com

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
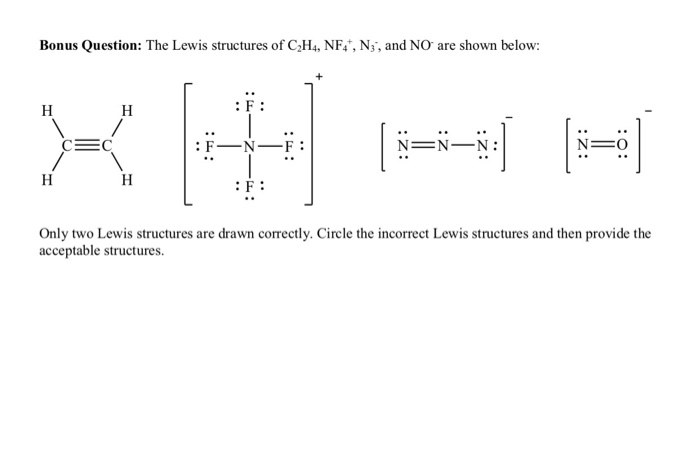
Bonus Question The Lewis Structures Of C2h4 Nf N Chegg Com

Draw The Lewis Structure For The C2h4 Ske Clutch Prep

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist

C2h4 Lewis Structure C2h4 Lewis Structure Molecular Geometry
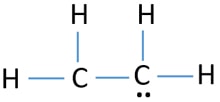
Ethene C2h4 Lewis Structure Hybridization

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist

Draw The Electron Dot Structure Of Ethene C2h4 Brainly In

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
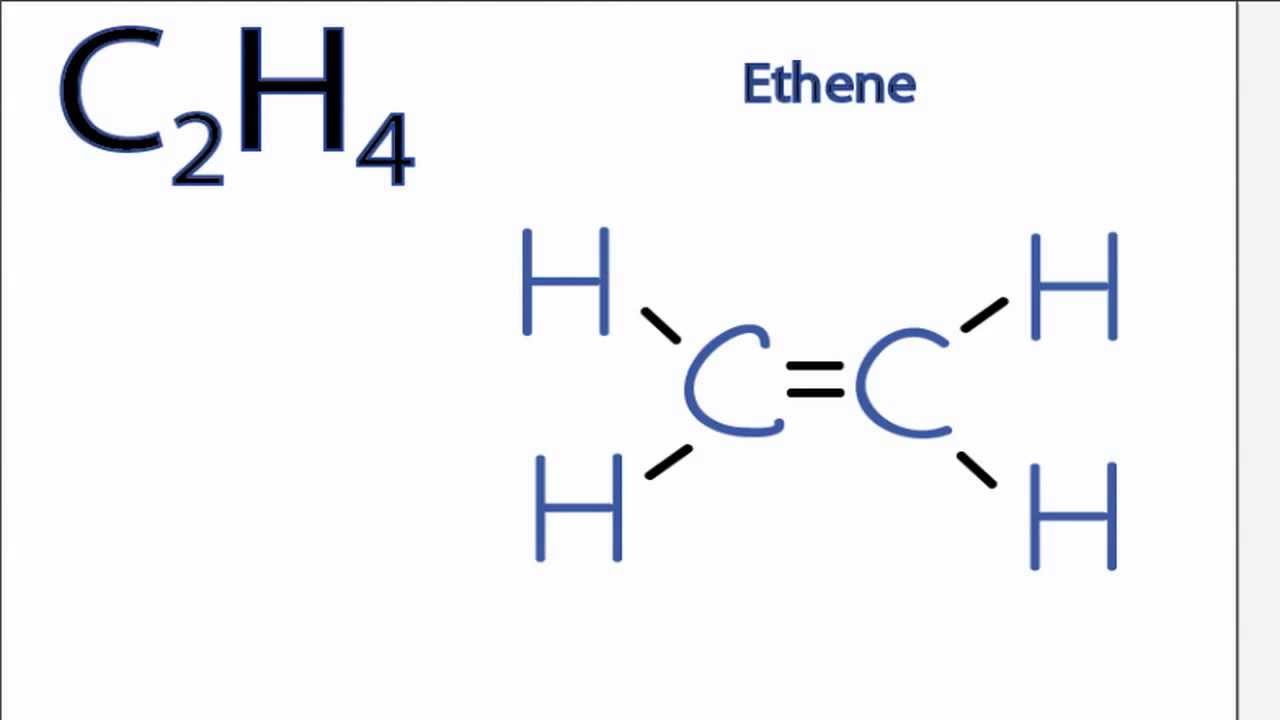
Is C2h4 Polar Or Nonpolar Youtube
Lewis Structure Of C2h4 Biochemhelp

C2h4 Lewis Structure Molecular Or Electron Geometry Polar Or Nonpolar

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
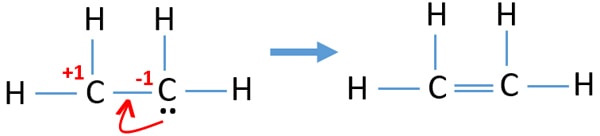
Ethene C2h4 Lewis Structure Hybridization
Lewis Electron Dot Structures Ck 12 Foundation
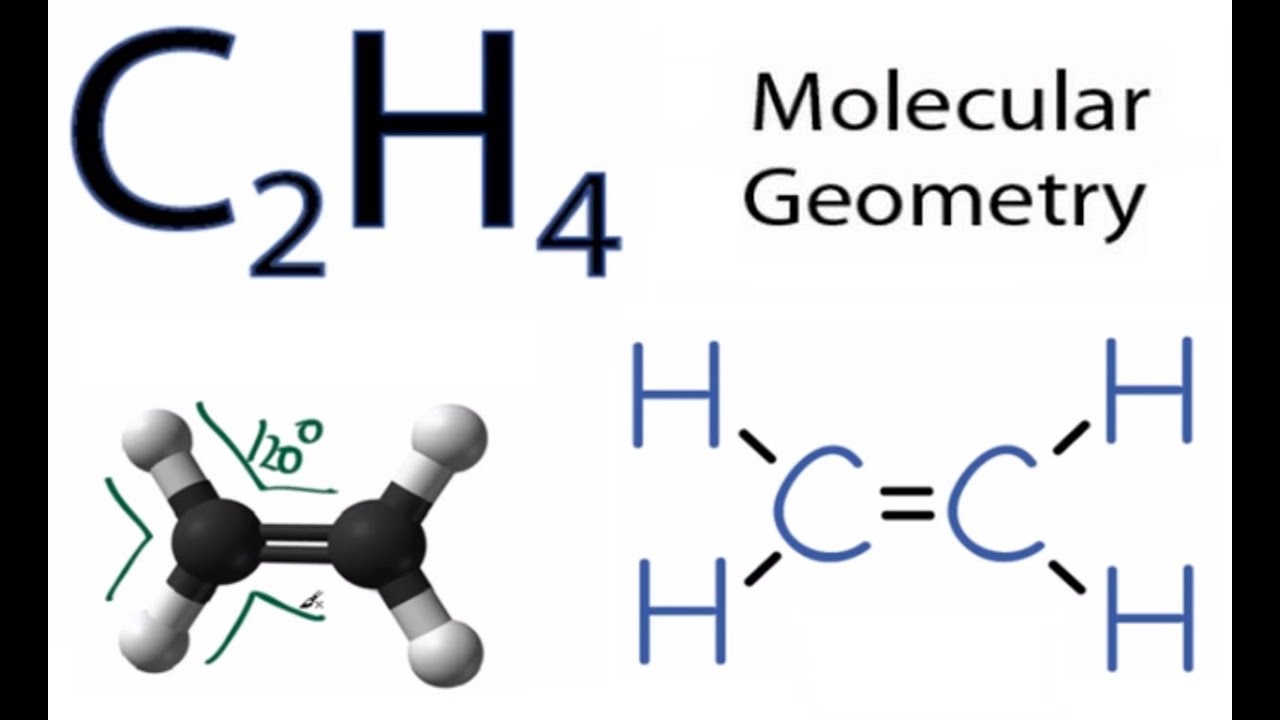
C2h4 Molecular Geometry Shape And Bond Angles Youtube
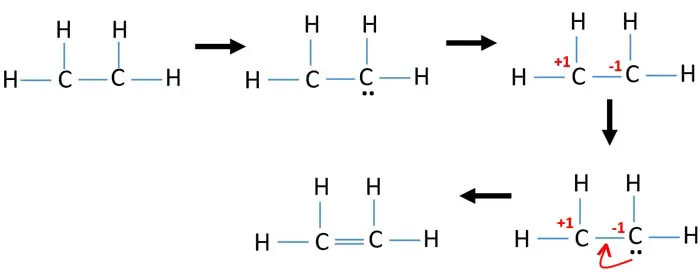
Ethene C2h4 Lewis Structure Hybridization
How Is C2h4 Planar While C2h6 Is Non Planar Quora
