N-3 Lewis Structure
You will every fact of drawing lewis structures from this tutorial which will help you to draw more lewis structures in the future. Steps of drawing the lewis structure of N 3- ion are explained in detail in this tutorial.

Lewis Structures Made Easy Examples And Tricks For Drawing Lewis Dot Diagrams Of Molecules Yo High School Chemistry Teaching Science Organic Chemistry Study
A step-by-step explanation of how to draw the N3- Lewis Dot Structure Azide ionFor the N3- structure use the periodic table to find the total number of va.

N-3 lewis structure. Lewis Structure for Azide Ion. But when there are. Therefore is it a stable structure.
NH3 Lewis structure molecular geometry. So it is also referred to by the name of nitrogen oxoanion. In the Lewis structure for NO2 the Nitrogen atom is the least electronegative atom and goes at the center of the structure.
In lewis structure of N 3- ion contains two NN bonds. A Lewis structure also known as Lewis dot structure is a simplified representation of symbols of the elements. Lone pairs unpaired electrons and single double or triple bonds are used to indicate where the valence electrons are located around each atom in a Lewis structure.
Ammonia NH 3 Lewis Structure Steps of Drawing. Most structuresespecially those containing second row. The Lewis structure of the tetra atomic ammonia NH3 molecule has three single sigma bonds between the nitrogen and the hydrogen atoms.
What is the Lewis structure drawing for N 3 N 3. N has tetrahedral electronic geometry. The formula of molecule is eqtextN_3- eq.
Organic or inorganic compounds that contain the -N3 group. In the lewis structure of ammonia NH 3 there are three N-H bonds and one lone pair on nitrogen atomLewis structure of NH 3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Valence electronic structures can be visualized by drawing Lewis symbols for atoms and monatomic ions and Lewis structures for molecules and polyatomic ions.
It is a reason why the bond angle is 107 where it should have been 1095. The compound has its chemical name as nitrate formed after nitric acid looses a proton from it. It has a role as a mitochondrial respiratory-chain inhibitor.
Total valence electrons of nitrogen and oxygen atoms and negative charge are considered to draw the NO 3-lewis structure. A step-by-step explanation of how to draw the N3- Lewis Dot StructureFor the N3- Lewis structure use the periodic table to find the total number of valence. Tetrahedral because its four groups of electrons.
Thus Ammonia is an example of the molecule during which the central. Because of this well try to get as close to an octet as we can on the central Nitrogen N atom. There are charges on all nitrogen atoms.
There are a total of 16 valence electrons in the N 3- Lewis structure. But when there are charges on lot of atoms and cannot reduce charges furthermore that structure is very unstable. For the N3- Lewis structure calculate the total number of valence electrons for the N3- molecule.
Each step of drawing the lewis structure of NH 3 is explained in detail in this tutorial. Moreover the presence of a single lone pair of electrons on the nitrogen atom is responsible for the bent geometrical structure of the NH3 molecule. Put brackets and a negative sign around the N 3- Lewis structure.
NO3 is a polyatomic ion with a negative charge. NO3 Lewis Structure Molecular Geometry and Hybridization. This chemistry video tutorial explains how to draw the lewis structure of the Ammonium Ion NH4My Website.
One group has an unshared pair of electrons. NH3 electron geometry is. Azide anion is a pseudohalide anion.
The NO2 Lewis structure has a total of 17 valence electrons. With N 3- youll need to form two double bonds between the Nitrogen atoms to fill the octets and still use only the 34 valence electrons available for the molecule. Lewis structure of NH 4 has a 1 charge on nitrogen atom.
First we should know atoms in a lewis structure can contain charges. Medical Subject Headings MeSH. In the Lewis Structure for N3- youll need to place a double bonds between the Nitrogen atoms to achieve full outer shells on all atoms while only using the valence electrons available for the molecule.
Therefore it requires three bonds to complete its octet. Nitrogen N belongs to group 15 and thus it has five valence electrons. Each outside nitrogen atoms have two lone pairs and center nitrogen atom does not have lone pairs.
Lewis Structure of NO 3-Nitrite ion Lewis structure of NO 3-ion is drawn step by step in this tutorial. Now lets move forward and realize the electron geometry. Besides this the.
Lewis Structure N3 Sodium azide Wikipedia Estructura de Lewis Enlace químico 4 Solved. When spread of charges around the ion or molecule is low that structure become more stable. Nitrate is an important source of nitrogen and oxygen.
This will mean that it will only have 7 valence electrons. It is a conjugate base of a hydrogen azide. O CHEMICAL BONDING Calculating Formal Charge A Stu.
Its not common to have an odd number of valence electrons in a Lewis structure.

How To Find Valence Electrons And Total Electrons Youtube Chemistry Lessons Teaching Chemistry Chemistry Help

Snc 2d Strand 3 Chemical Reactions Unit 3 Chemical Structure Chemical Structure Science Classroom High School Chemistry

Lewis Dot Structures Formula Units Ionic Formulas Clear Simple Chemistry Classroom Teaching Chemistry Science Chemistry

Lewis Structure For Co Carbon Monoxide In 2021 Molecules Lewis Carbon Monoxide

N3 Lewis Structure Azide Ion In 2021 Math Equations Lewis Molecules

No3 Lewis Structure How To Draw The Lewis Structure For No3 Chemistry Science Chemistry Chemistry Help

Lewis Structures Made Easy Examples And Tricks For Drawing Lewis Dot Diagrams Of Molecules Yo High School Chemistry Teaching Science Organic Chemistry Study

Nh4 Lewis Structure How To Draw The Dot Structure For Nh4 Ammonium Science Chemistry Molecular Geometry Chemistry
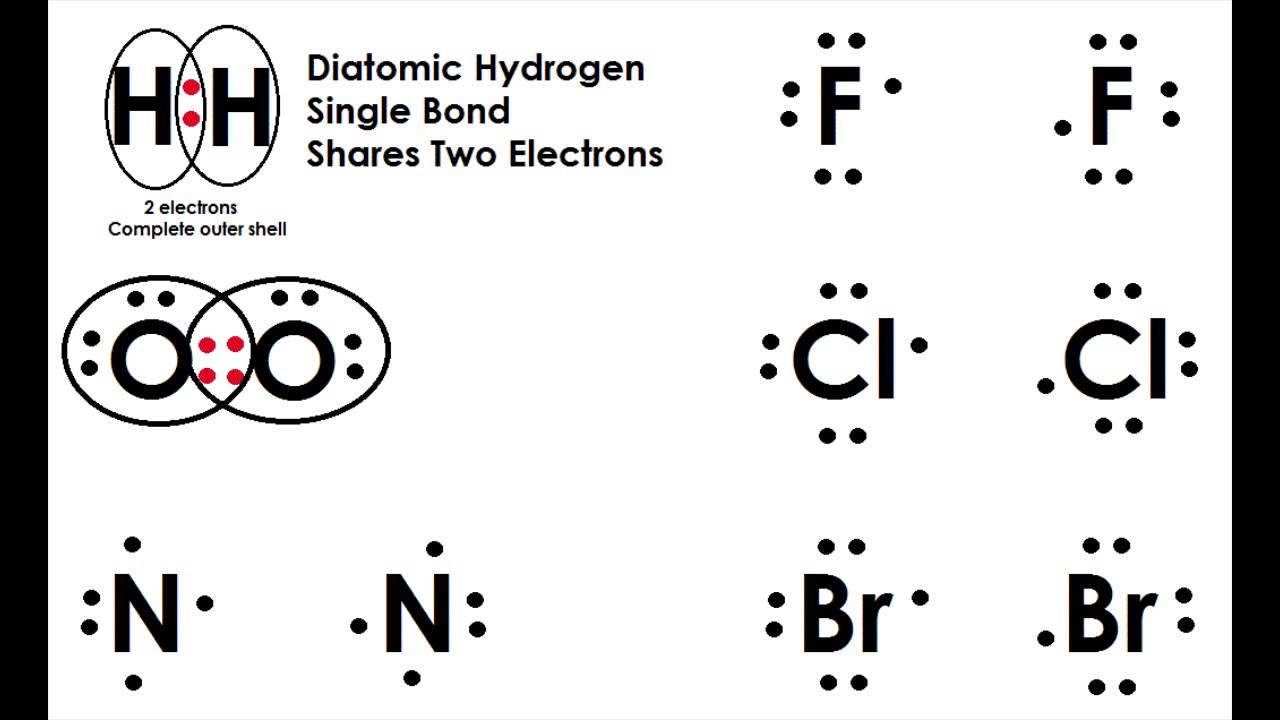
Diatomic Molecules And Covalent Bonding Covalent Bonding Molecules Bond

Ocn Lewis Structure How To Draw The Lewis Structure For Ocn Lewis Tech Company Logos Drawings

Oxygen Electron Configuration How To Write The Electron Configuration For Oxygen O In 2021 Electron Configuration Electrons Oxygen

3 Membered Ring Nitrogen Heterocycles Made By Roman A Valiulin With Chemdraw Chemistry Notes Nitrogen Chemistry

Calculating No3 Formal Charges Calculating Formal Charges For No3 Chemistry Classroom Science Chemistry Chemistry

Drawing A Lewis Structure For Dinitrogen Monoxide Nitrous Oxide N2o Linked To Lewis Structures How Tos

Sf4 Lewis Structure How To Draw The Lewis Structure For Sf4 Teaching Chemistry Chemistry Worksheets Chemistry Education

Trigonal Pyramid Molecular Geometry Molecular Geometry Molecular Shapes Chemistry Projects

Simple Procedure For Writing Lewis Structures For Methanimine Ch2nh 25 Chemistry Net Writing Chemistry Math Equations

Simple Method For Writing Lewis Structures For N2o3 Molecular Geometry Chemistry Help Molecular Shapes
