Lewis Dot Structure Sef4
The dots in a Lewis dot structure represent an atoms valence electrons and the placement of the dots indicate how the electrons are distributed in a molecule. Molar mass Cu 6355g mol.

Sef4 Lewis Structure How To Draw The Lewis Structure For Sef4 Youtube
Quiz your students on lewis dot diagram structure for sef4 molecular geometry hybridization.

Lewis dot structure sef4. Note that Se can hold more than eight valence electronsFor the SeF6 Lewis structure. So the final lewis structure of BrO3-contains two double bonds and one single bond each oxygen atom has 8 valence electrons and the bromine central atom has 12 valence electrons in its outermost shell as it has the ability to expand octet. It has one lone pair and four bonds around sulfur.
A step-by-step explanation of how to draw the SeF4 Lewis Dot Structure Selenium TetrafluorideFor the SeF4 structure use the periodic table to find the tot. The shape is like a seesaw. The way to determine the molecular geometry of SeF4 is to first draw the Lewis Dot Structure.
XeF4 Lewis Structure - How to Draw the Lewis Structure. SeF 4 is Lewis structure with Selenium which can hold more than 8 valence electrons. Its a good idea to check the formal charges for your SeF 4 Lewis structure.
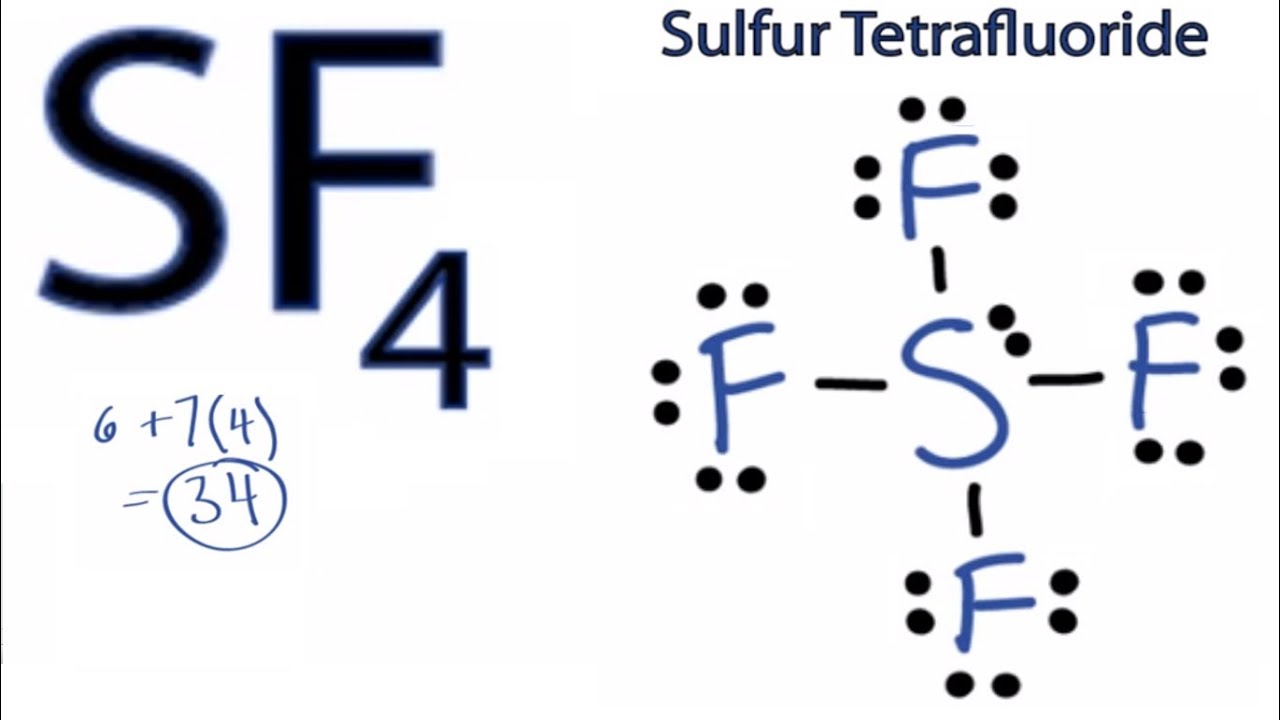
Lewis structure is a pictorial representation of the bonds and valence electrons in the molecule. SeF 4 is Lewis structure with Selenium which can hold more than 8 valence electrons. Draw the correct Lewis structure of SF4.
SeF4 lewis structure is made up of one selenium and four fluorine atoms selenium is the central atom and fluorine is kept outside in the lewis diagram. The Lewis structure of SF4 is the combination of 34 valence electron and 5 electron pairs around the Sulfur in which there are four bonding pairs and one lone pair. The overall charge in BrO3- is -1.
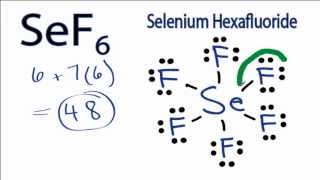
Complete the Lewis structure of serine. This compound is generally identified as being a colorless gas. A step-by-step explanation of how to draw the SeF6 Lewis Structure.
Since there are seven fluorine f atoms it will be necessary. Since there are seven Fluorine F atoms it will be necessary. Bond dipoles do not cancel.
SF4 Lewis Structure Molecular Geometry Hybridization and MO Diagram - Techiescientist SF4 Lewis Structure Molecular Geometry Hybridization and MO Diagram SF4 or sulfur tetrafluoride is a compound that has a distinct odor of sulfur or rotten eggs. A Draw the Lewis dot structure for the molecule by placing atoms on the grid and connecting them with bonds. Silicon tetrafluoride SiF4 or F4Si CID 24556 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more.
Youll have a pair of electrons left over after filling octets of the F atoms. The valence electrons that participate in forming bonds are called bonding pairs of electrons whereas the electrons that do not participate or form any. Vapor is heavier than air.
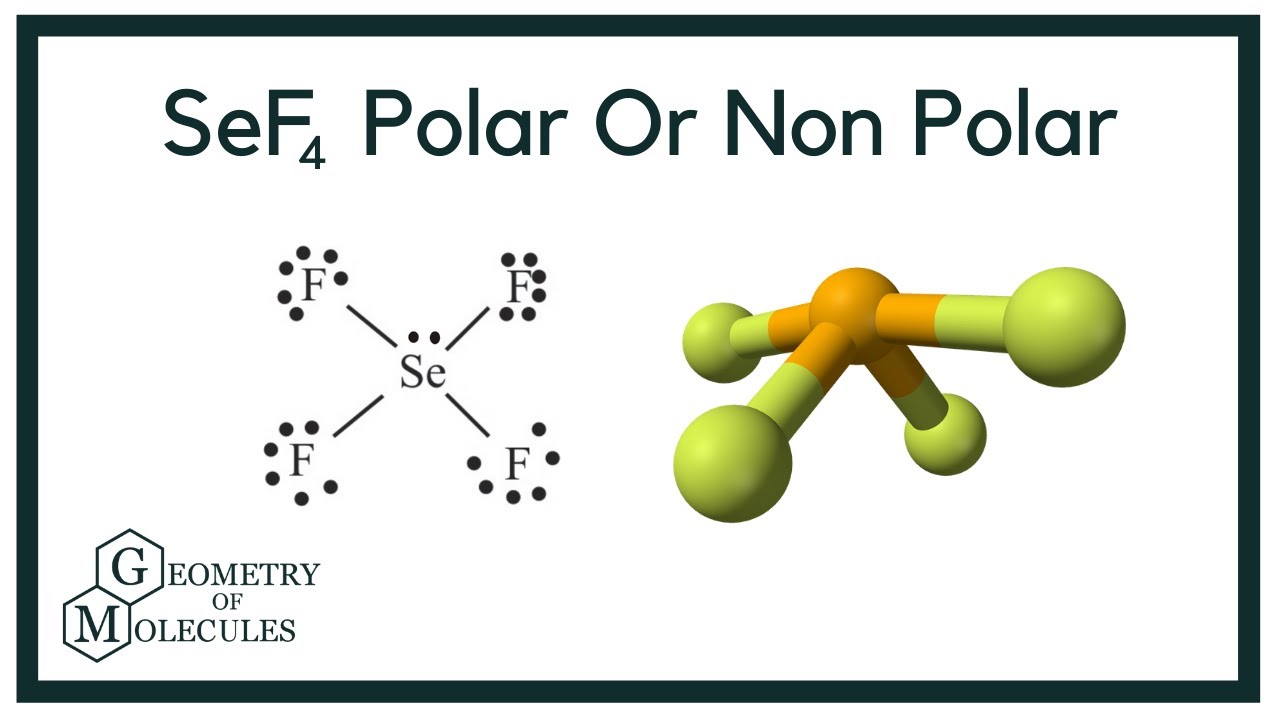
There is one lone pair present on the central atom in the SeF4 lewis structure and 12 lone pairs on outer atoms. A Lewis dot structure illustrates the sharing of electrons between atoms in covalent or polar covalent bonds both explained in this page of my website. Use VSEPR to find the correct geometry for an atom with five groups around it trigonal bipyramid.
Since there are seven Fluorine F atoms it will be necessary. You can put these on the central Se atom. SeF4 is Lewis structure with Selenium which can hold more than 8 valence electrons.
Name The Molecular Geometry Name The Electron Main. You can put these on the central Se atom. This electron arrangement is known as Trigonal Bipyramidal.
Youll have a pair of electrons left over after filling octets of the F atoms. The skeletal structures of serine is shown below. The Lewis structure for SeF 4 has 34 valence electrons available to work with.
The bonds formed between two atoms are depicted using lines whereas the valence electrons not forming any bonds are shown by dots. Since there are seven Fluorine F atoms it will be necessary. Quiz your students on Lewis Dot Diagram Structure For SeF4 Molecular Geometry Hybridization Polar or Nonpolar using our fun classroom quiz game Quizalize and personalize your teaching.
Try to draw the xef 4 lewis structure before watching the video. Include all hydrogen atoms and nonbonding. Also know what is the Lewis structure for SeF4.
Draw The Lewis Dot Structure Of SeF4 And.
What Is The Molecular Geometry Of Sef4 How Is It Determined Quora
Lewis Structures And Shapes Of Molecules Teaching Resources
Sef6 Lewis Structure How To Draw The Lewis Structure For Selenium Hexafluoride Youtube
Sf4 Lewis Structure How To Draw The Lewis Structure For Sf4 Youtube

File Selenium Tetrafluoride Svg Wikimedia Commons

Write Lewis Structures For Sef4 And Sef6 Is The Octet Rule Satisfied For Se Study Com
Is Sef4 Polar Or Nonpolar Quora

Sef4 Lewis Structure Molecular Geometry Polar Or Nonpolar Bond Angle

How To Calculate The Formal Charges For Sf4 Sulfur Tetrafluoride Youtube

Consider The Molecule Sef4 A Draw The Lewis Structure B What Is The Hybridization Of Se C What Is The Electron Geometry D What Is The Molecular Geometry E What Degree Angles
Is Sef4 Polar Or Non Polar Selenium Tetrafluoride Youtube
For Sef4 Write The Lewis Structure Showing All Chegg Com

Draw The Lewis Structure Of Selenium Tetralfluoride Sef4 Clutch Prep

Sef4 Lewis Structure How To Draw The Lewis Structure For Sef4 Youtube
Http Www Msubillings Edu Sciencefaculty Handouts Wiles Chem 20116 Solution 20set 202 Pdf

What Is The Molecular Geometry Of Sef4 How Is It Determined Quora

Determine The Molecular Geometry Of Sef4 Clutch Prep



