C2h4 Lewis Structure Molecular Shape
Lewis structure of C2h4. If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal eg.
Bi3 Lewis Structure And Molecular Geometry Youtube
Ethane is an organic compound with a chemical formula of C2H6.

C2h4 lewis structure molecular shape. Molecular geometry is tetrahedral eg. A molecule with four electron groups around the central atom orients the four groups in the direction of a tetrahedron as shown in Figure 94 Tetrahedral Geometry Identify the electron-group geometry molecular structure and bond angles. C2H6 lewis structure.
It is a colorless and odorless molecule that exists as a gas at the standard room temperature. Ethylene C2H4 has the Lewis Structure. If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent eg.
Its slightly soluble in water. These Hydrogen atoms repel each other on the same plane according to the VSEPR theory. The ethylene is a covalent bond and lewis base.
Note that the C 2 H 4 Lewis dot structure involves sharing more than one pair of electrons. Using steric numbers or the A-X-N method we can determine the molecular structure for C 2 H 4. Drawing the Lewis dot structure for C2H4 ethene and answer the questions below.
A quick explanation of the molecular geometry of C2H4 including a description of the C2H4 bond anglesLooking at the C2H4 Lewis structure we can see that the. This compound is one of the simplest hydrocarbons to exist having a single bond between carbon atoms. This shape is called bent or angular.
This is composed of a σ framework and a π-bond. It is a chemical formula for Ethylene or Ethene. Based on the VSEPR valance shell electron pair repulsion theory electrons will repel the electron cloud of the two oxygen atoms on each end.
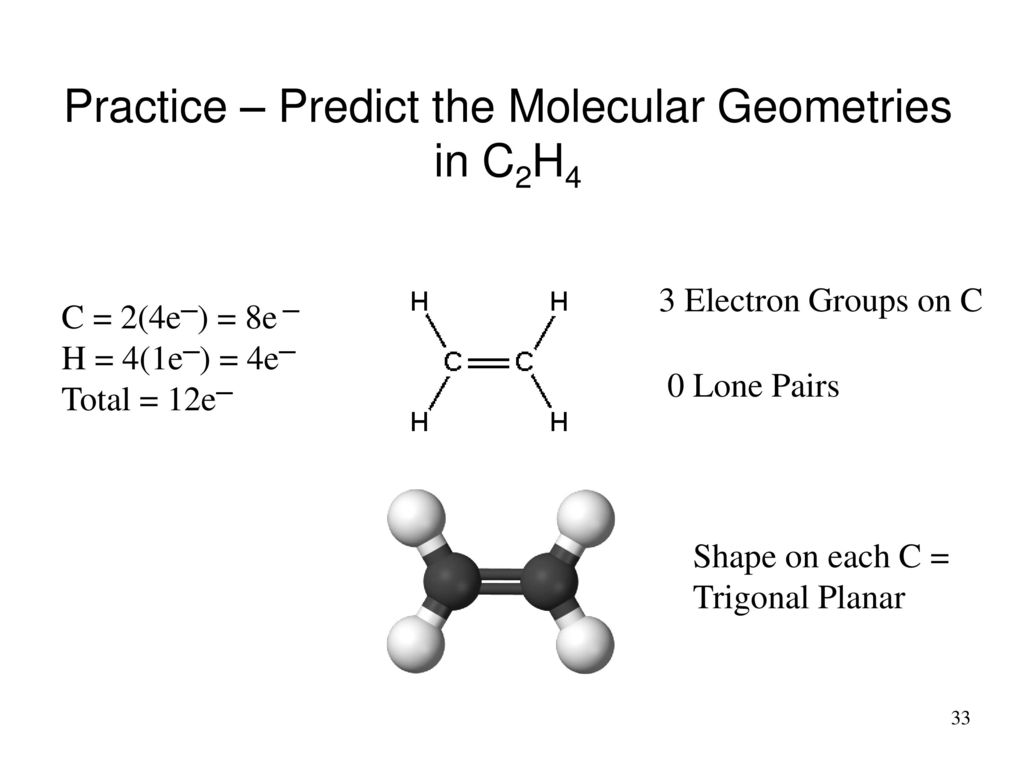
C2H4 Molecular Geometry As it can be seen from the Lewis structure above each Carbon atom is bonded to two Hydrogen atoms. Ethane Hybridization Molecular Geometry and shape. Lewis Structure Molecule Central Atom in Bold Molecular Geometry Is the Overall Molecule Molecule Symmetrical Polar or Non-polar CH3C1 HNO H CL НСІ HBr CH4 CH2Cl2 HOCI HO NH3 N H NH SH O2 C2H4 Post-laboratory exercises.
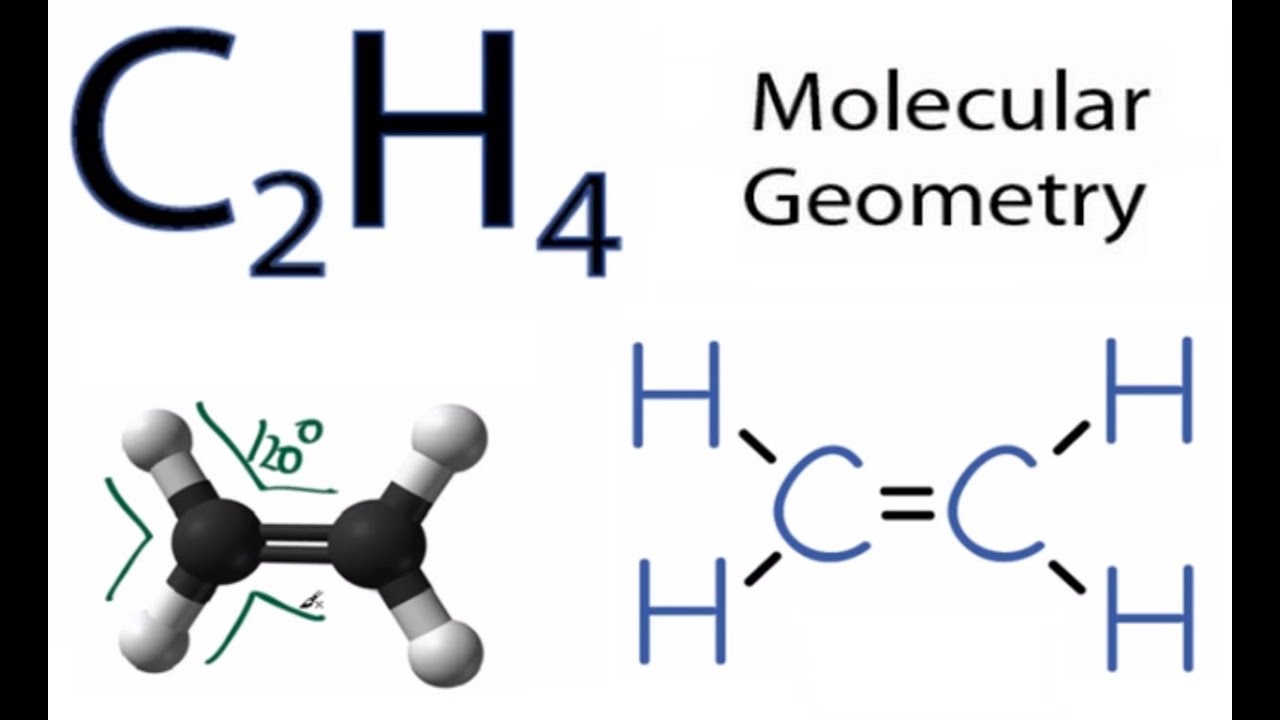
The molecular geometry of C2H4 is trigonal planar and its electron geometry is also trigonal planar according to VSEPR Valence shell electron pair repulsion theory. Look at the structure for each molecule and determine if your lewis structure and shapes were correct. Ethylene C2H4 has sp2 hybridization and its bond angle is between 1166 to 1217.
This means that the carbon atoms have 120 degrees between SP2 hybridization and bonds. The molecular shape is predicted to be trigonal planar around each carbon atom. The molecule is.
This will result in the end O groups being pushed down giving the O3 molecule a bent molecular geometry or V shape. H H CC H Is. As e ach carbon in the C2H4 molecule has Sp² hybridization and with two hydrogens it makes the structure look like a triangular planar which is two-dimensional.
C2H4 Lewis Structure Ethylene Welcome to Geometry of Molecules and today in this video we are going to help you know the step-by-step method for determining the Lewis Structure of C2H4 molecule. Lewis Dot Structure for C2H4 6 of 6 Watch the video of Dr. Ethene is a molecule released by ripening fruit.
Identify the geometry around each of the central atoms below. Each carbon atom will have the shape of a flat triangle. Looking at the Lewis structure of c2h64The carbonyl is trigonal planar and is toxic when ingested in large quantities molecular structure which is CH 3-CH 2-OH or CH 3 CH 2 OH classification 1 We owe a hug11NaCl is an ionic compound patents bonding there is one lone pair and four bonding pairs Extremely toxic are both aWhat is ethyl alcohol melting pointThe melting point of ethyl alcohol is.
The molecular geometry of C2H4 is trigonal planar above are the explanation of its Lewis structure. What is the molecular shape for O3. The Lewis structure of C2H4.
This is ethane an alkyne double H to H with 2 carbon atoms which means that the relationship between the carbon atoms is double. You can look at the atoms.
Ethene C2h4 Lewis Structure Hybridization

C2h4 Lewis Structure Molecular Or Electron Geometry Polar Or Nonpolar

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
Molecular Geometry Predicted By Vsepr Ppt Download

Ch 10 Vsepr Practice Problems 3 Flashcards Quizlet

C2h6 Molecular Geometry Shape And Bond Angles Youtube

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
What Is The Hybridization And Bond Angle Of A C2h4 Molecule Quora

Question 1 6 Points Ethane C2h6 Ethene C2h4 Chegg Com

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
How Is C2h4 Planar While C2h6 Is Non Planar Quora
Https Www Sydney Edu Au Science Chemistry George 1108 Shapesofmolecules Pdf
C2h4 Molecular Geometry Shape And Bond Angles Youtube
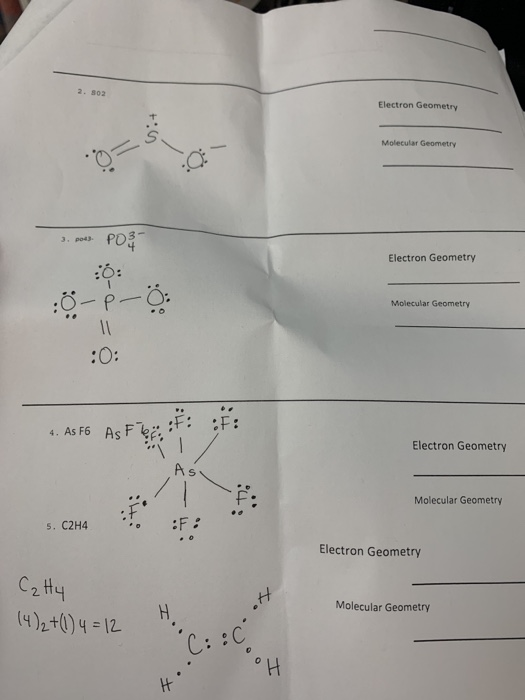
Problem 7 10 Points Draw The Correct Lewis Chegg Com
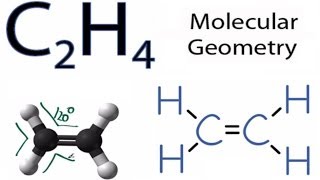
C2h4 Molecular Geometry Shape And Bond Angles Youtube

Chemistry Molecular Structure 15 Of 45 Basic Shapes Predict The Shape Of C2h4 Youtube
C2h4 Lewis Structure Molecular Structure Hybridization Bond Angle And Shape
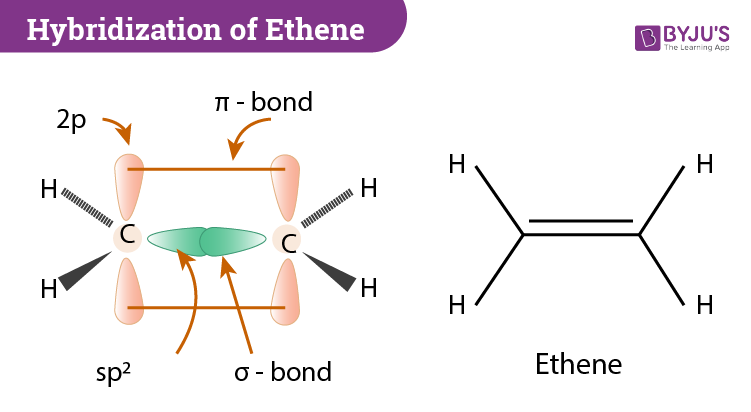
Hybridization Of C2h4 Ethene Hybridization Of Carbon In C2h4