Hno3 Lewis Structure Formal Charge
H3O Lewis Structure. 1 Draw the Lewis structure of nitric acid HNO 3 that minimizes formal chargesAssign lone pairs radical electrons and atomic charges where appropriate 2 Calculate the electrons required.

Formal Charge And Its Properties Formal Charge Equation Importance
In order to calculate the formal charges for HNO3 well use the equationFormal charge of valence electrons - nonbonding val electrons - bonding ele.

Hno3 lewis structure formal charge. Both sulfuric and nitric acids are NEUTRAL species. Lewis structure of nitric acid There is a NO bond in nitric acid lewis structure. First of all we need to calculate the total number of valence electrons present in hydronium ion.
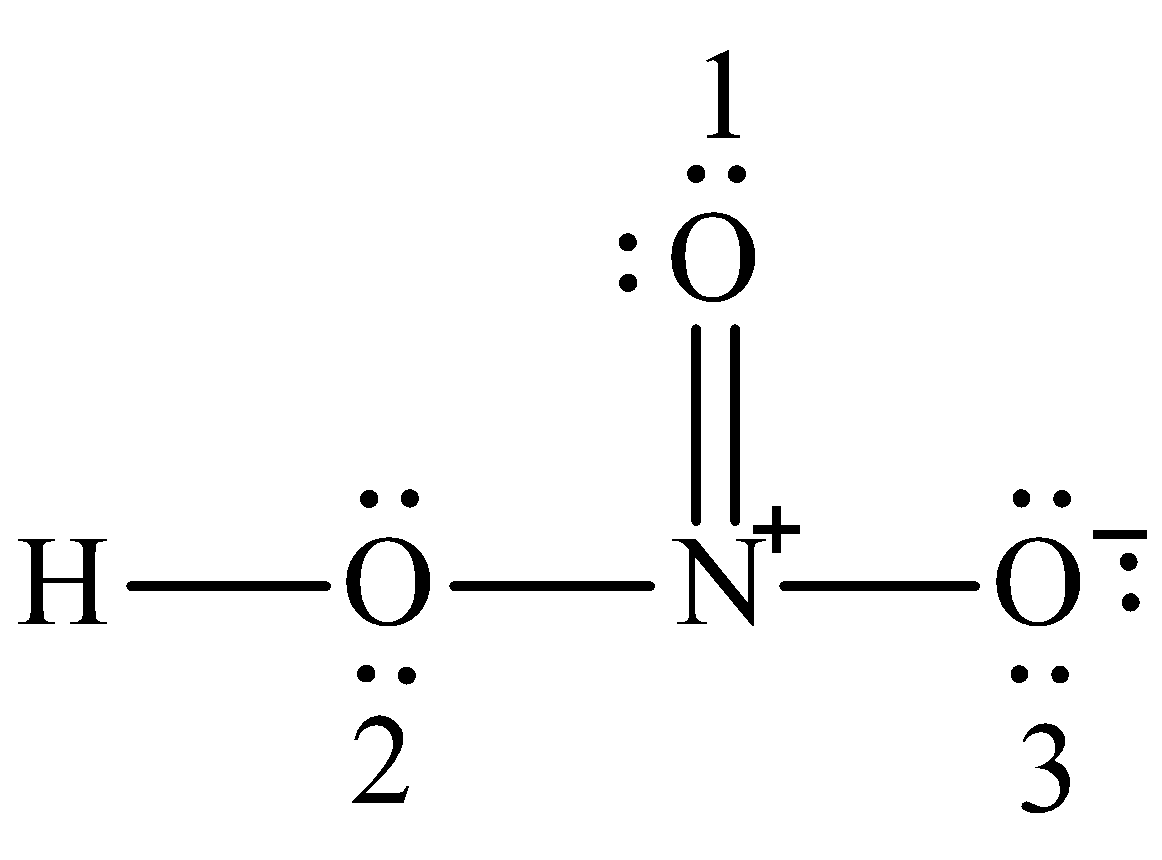
Drag the appropriate labels to their respective targets. Home Draw the lewis dot structure of H2SO4 show formal charges. In the lewis structure of nitric acid there is a 1 charge on nitrogen atom and one double bond between nitrogen and one oxygen atom.
Lone Pair e- Q Bond Pair e- 18 4 14. In the previous video we saw some steps for drawing dot structures in this video were going to use those same steps to draw a few more dot structures but were also going to talk about how formal charge relates to dot structures so well get back to this definition in a minute for right now lets draw a quick dot structure for the ammonium cation so NH 4 plus the first thing you do is find. For the HNO3 Lewis structure calculate the total number of valence electrons for the HNO3 molecule.
Of course the molecule is neutral and the Lewis structure reflects this. In this structure the central atom is nitrogen because it is in least number. Show how its stronger acid than HNO3.
Based on octet rule alone there are 3 possible resonance structures that are favorable. Hydrogen 1 3Hydrogen 3 Oxygen 6 Total 9. Lewis dot formulae should and do reflect this neutrality.
Draw the lewis dot structure of H2SO4 show formal charges. View Available Hints ResetHelp. HNO 3 Nitric acid lewis stricture is drawn step by step by using valence electrons of each element.
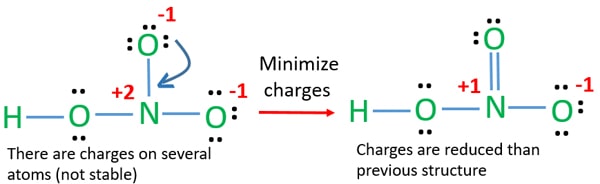
In the Lewis structures of N F 3 and C O 3 2 nitrogen and carbon occupy the central position whereas fluorine and oxygen occupy the terminal positions. A reasonable Lewis structure is H O N OO. However the first two resonance structures are significantly more favorable than the third because they have smaller amount of formal charges.
Lewis Structure Formal charges Compound SCN SO42- H2SO4 NCO NO. This is a pattern seen with many acids. The nitrogen in HNO3 has a formal 1 charge and a formal double bond to one of its oxygens.
Now the important point is not to forget about the sign. Check the formal charges to be sure that each atom has a formal charge of zero. Lewis structure of NO 3 1- after assigning the charge the net charge will be -1.
H2SO3 HNO3 HCIO3 Compound Lewis Structure Formal Charge HBr CCl4 CHO OF 2 CO2 BCI. A step-by-step explanation of how to draw the HNO3 Lewis Structure Nitric Acid. Labels can be used once more than once or not at all.
Remember the definition of a Lewis acid. Reason In Lewis representation the least electronegative atom occupies the central position in the moleculeion. Sign indicates losing an electron from the total valence electrons.
The structure of NO 3 1- is. Drawing the Lewis Structure for HNO 3. Make the bonds between central atom and corner atoms.
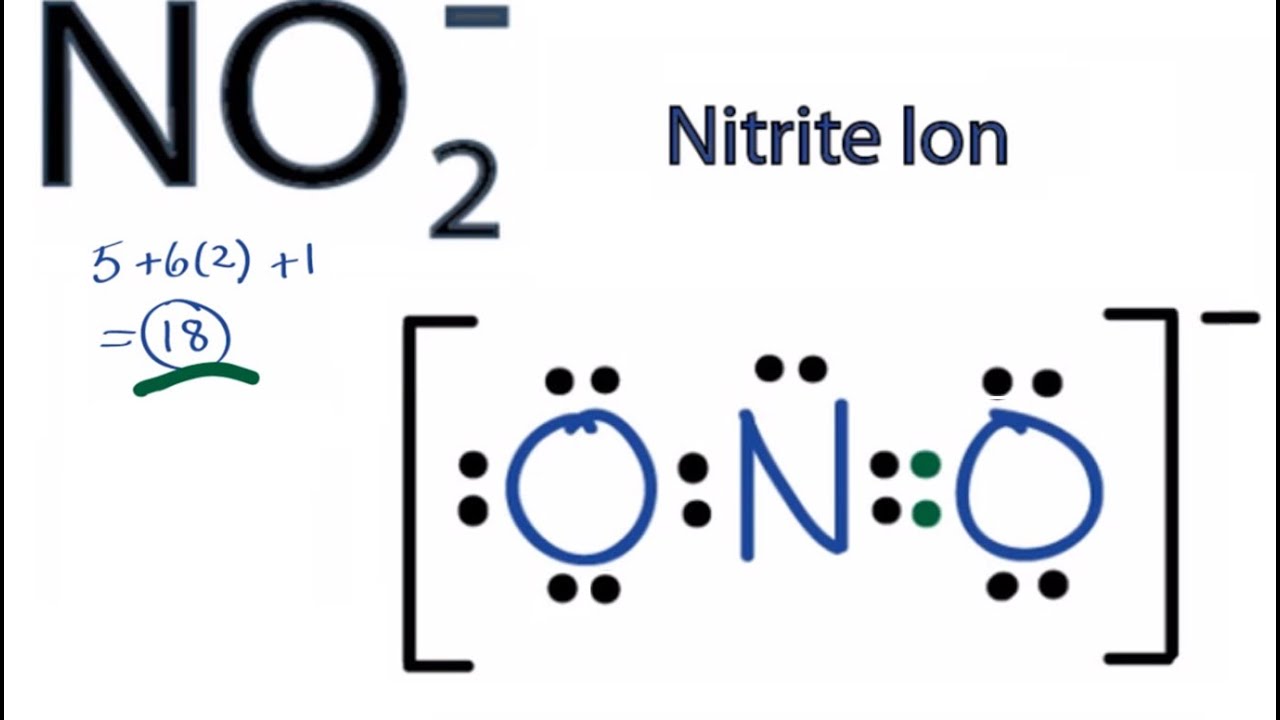
Part A Write formal charges for each atom in nitric acid HNO3 which has the Lewis structure shown. Lewis Structure of NO 2 1-Q 5 2 x 6 1 18. The HNO3 Lewis structure is best thought of as the NO3 with an H attache.
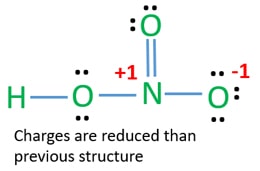
The oxygen atoms are marked with letters to distinguish them from each other H-O-N-O. In HNO 3 Lewis structure Nitrogen N is the least electronegative atom and goes in the center of the Lewis structure. Formal charge on an atom in a Lewis structure total number of valence electrons in free atom total number of non-bonding lone pairs electrons 12 total number of bonding or shared electrons.
In reality the bond order is more complex But the nitrogen can accept an electron pair donating its formal pi bond to the oxygen that is formally double bonded to it. The HNO3 Lewis structure is best thought of as the NO3 with an H attached to one of the oxygen atoms. After determining how many valence electrons there are in HNO3 place them around the central atom to.
In this case the formal charges will be closer to zero if you place a double bond beteween the Nitrogen atom and the Oxygen atom without the H attached. Anything that can accept an electron pair. Lets try to draw the lewis structure of H3O.
BCIF CHCI3 This problem has been solved. Show how its stronger acid than HNO3. For HNO3 in order to satisfy the octet rule the nitrogen atom would form 1 double bond and 2 single bonds.
What is the formal charge on each atom in HNO3 and the Lewis structure for it. What is the formal charge on each atom in PH4 nd the Lewis structure for it. What is the formal charge on each atom in CH3Cl and the Lewis structure for it.
One of the oxygen atoms has a formal negative charge and the nitrogen atom is quaternized and bears a formal positive charge. Bone Pair e- 4.
Hno3 Nitric Acid Lewis Structure

The Lewis Structure Of Hno3 Chemistry Stack Exchange

Nitric Acid Hno3 Lewis Structure Properties Uses Rankred

1 Draw The Lewis Structure Of Nitric Acid Hno 3 That Minimizes Formal Charges Assign Lone Pairs Radical Electrons And Atomic Charges Where Appropriate 2 Calculate The Electrons Required Er Study Com

Consider The Lewis Structure For The Nitri Clutch Prep

126 Hso4 Lewis Structure How To Draw The Lewis Structure For The Bisulfate Ion Youtube Science Chemistry Chemistry Organic Chemistry

How Is The Lewis Dot Structure For Hno3 Determined Quora
Write Lewis Structure Of The Following Compounds And Show Formal Charge On Each Atom Hno3 No2 H2so4 Sarthaks Econnect Largest Online Education Community

Which Of The Following Are Acceptable Lewis Chegg Com

Oneclass Write Formal Charges For Each Atom In Nitric Acid Hno3 Which Has The Lewis Structure Show
Hno3 Nitric Acid Lewis Structure
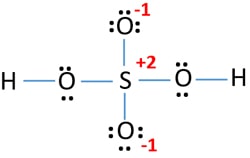
Lewis Structure Of Sulfuric Acid H2so4 Steps Of Drawing

Draw A Lewis Structure For Nitric Acid The Hydrogen Atom Is Attached To One Of The Oxygen Atoms Brainly Com

Pcl3 Lewis Structure Phosphorus Trichloride In 2021 Math Equations Lewis Chemical Formula

Pcl3 Lewis Structure Phosphorus Trichloride In 2021 Math Equations Lewis Chemical Formula
4 2 Lewis Structures Problems Chemistry Libretexts
Write Lewis Structure Of The Hno3 And Show Formal Charge Class 12 Chemistry Cbse