Lewis Structure For Hono2(hno3)
Lewis structure of nitrous acid. However the first two resonance structures are significantly more favorable than the third because they have smaller amount of formal charges.
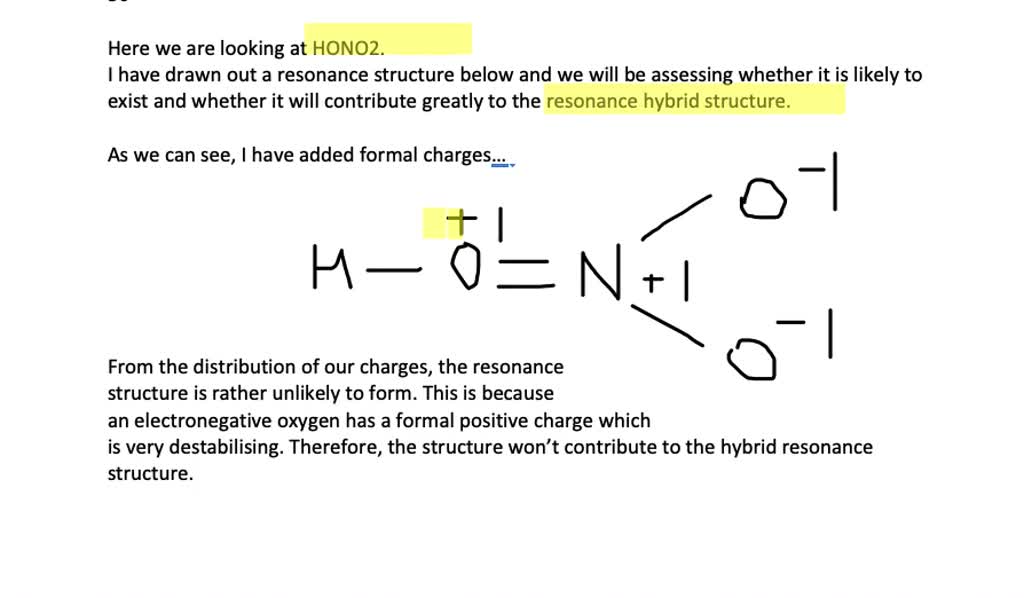
Solved The Lewis Structure Of Nitric Acid Mathrm Hono 2 Is A Resonance Hybrid How Important Do You Think The Con
The HNO 3 Lewis structure is easier to think of if you consider it NO 3 with an H bonded to one of the oxygen atoms.

Lewis structure for hono2(hno3). In the lewis structure of nitric acid there is a 1 charge on nitrogen atom and one double bond between nitrogen and one oxygen atom. So we can put our Nitrogen in the center. What Is The Lewis Structure For HONO2HNO3.
Nitric acid Lewis Structure. Draw Lewis structure for one important resonance form of HNO3 HONO2. Its not common to have an odd number of valence electrons in a Lewis structure.
HNO3 HONO2 See all problems in Resonance Structures. For HNO3 in order to satisfy the octet rule the nitrogen atom would form 1 double bond and 2 single bonds. The HNO3 Lewis structure is best thought of as the NO3 with an H attache.
For the HNO3 Lewis structure calculate the total number of valence electrons for the HNO3 molecule. Or if you need more Lewis Structure practice you can also practice Lewis Structure practice problems. In the Lewis structure for NO2 the Nitrogen atom is the least electronegative atom and goes at the center of the structure.
Nitrogen atom is the center atom in HNO2. This is a pattern seen with many acids. Draw Lewis structures of all the important resonance forms of b HAsO42 HOAsO32.
Check the formal charges to be sure that each atom has a. After determining how many valence electrons there are in HNO3 place them around the central atom to complete the octets. 99 88 ratings Problem Details.
Steps of drawing lewis structure of HNO3. A step-by-step explanation of how to draw the HNO3 Lewis Structure Nitric Acid. None of the atoms has any formal charge.
Include all lone pair electrons. Include all lone pair electrons. Postby 705170809 Tue Nov 20 2018 1107 pm.
Write a Lewis structure for each of the following. The Lewis dot structure for HNO 3 nitric acid shows how the valence electrons are used in the bonding between atoms. Write a Lewis structure for each of the following.
So H1 N5 O6 if we sum it up we get 153624. Like in any lewis structure the first thing we do is calculate the valence electrons. The NO2 Lewis structure has a total of 17 valence electrons.
Based on octet rule alone there are 3 possible resonance structures that are favorable. Draw an appropriate Lewis structure nitric acid HNO which has structure HONO2. What is the Lewis structure for HONO2HNO3.
Because of this well try to get as close to an octet as we can on the central Nitrogen N atom. When we have an H in the front of a polyatomic ion like NO2 or SO3 we know that its going to be an acid and that the Hydrogen is going to go on the outside of one of the Oxygen. This will mean that it will only have 7 valence electrons.
HNO 3 Nitric acid lewis stricture is drawn step by step by using valence electrons of each element. Find step-by-step Chemistry solutions and your answer to the following textbook question. There are one CO bond one C-O bond and one O-H in HNO2lewis structure.
Lewis Structure for NO3-. The Lewis Dot Structure for HNO 3. How is the Lewis dot structure for HNO3 determined.
Draw Lewis structure for one important resonance form of HNO3 HONO2. Which of the following statements is true. This problem has been solved.
The HNO3 Lewis structure is best thought of as the NO3 with an H attached to one of the oxygen atoms. There are some steps to follow to draw lewis structures. D HONO2 HNO3 FREE Expert Solution.
In HNO 3 Lewis structure Nitrogen N is the least electronegative atom and goes in the center of the Lewis structure. Nitric acid is a strong oxidizing agent. The Lewis Structure of HAsO 4 2-94 292 ratings Problem Details.
This is the HNO2 Lewis structure. Draw Lewis structures of all the important resonance forms of a HNO3 HONO2. Since NO3- has resonance there are three different most stable structures but all of.
NO3- has resonance which allows all the bond angles to be the same between the different resonance structures since resonance doesnt move the actual formation of the atoms but only rearranges the double bonds. Heshan Nipuna last update.

Chemistry Chemical Bonding 19 Of 35 Lewis Structures Nitric Acid Hno3 Youtube

The Lewis Structure Of Hno3 Chemistry Stack Exchange

Hno3 Lewis Structure Nitric Acid Youtube
The Lewis Structure Of Hno3 Chemistry Stack Exchange
4 7 Molecular Structure And Polarity General Chemistry 1 2

Click The Draw Structure Button To Launch The Chegg Com

Lewis Structure Of Hono2 Hno3 Brainly Com
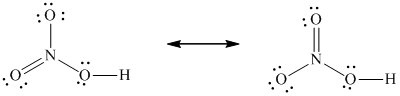
Solved Draw Lewis Structures Of All The Important Resonance Forms Chegg Com

Chemistry Chemical Bonding 19 Of 35 Lewis Structures Nitric Acid Hno3 Youtube
1 Write The Lewis Structures For The Following Chegg Com

Draw The Lewis Structure For Hno3 And State Its Molecular Geometry Is It Polar Or Nonpolar Study Com

The Lewis Structure Of Hno3 Chemistry Stack Exchange

Answer Write A Lewis Structure For Each O Clutch Prep

What Is The Action Of Hno3 On Ethane Quora

Draw Lewis Structures Of All The Important Clutch Prep

Raney Chemistry Chem Ii Dc Review Bonding
E None Of The Above 2 Write A Lewis Structure For Chegg Com


