Is Pf5 Polar
It is true that the P-F bond is polar because fluorine is more electronegative than phosphorous. But the dipole of the P-F bonds cancels out each other resulting in a zero net dipole moment.

Xef2 Lewis Structure Xenon Difluoride In 2021 Lewis Molecules Math Equations
Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.
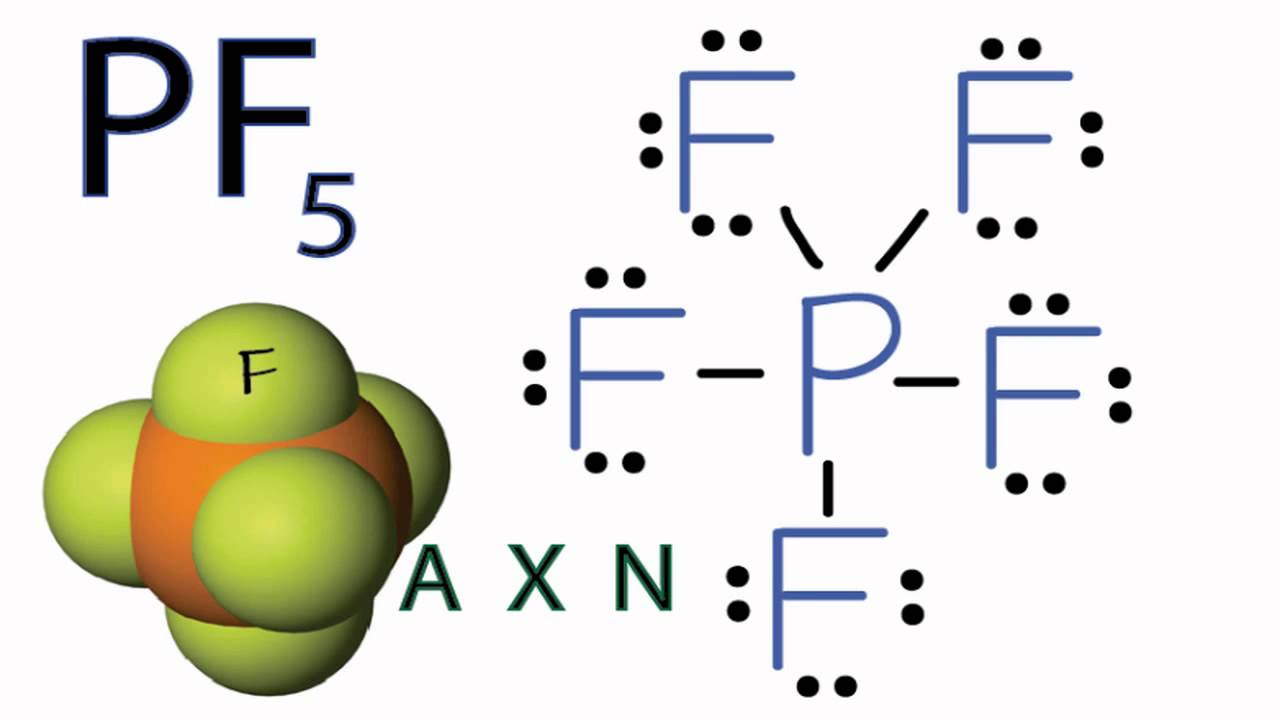
Is pf5 polar. No PF5 is not polar. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms. Answer PF5 Pentafluorophosphorane is Nonpolar What is polar and non-polar.
Answer PF5 Pentafluorophosphorane is Nonpolar What is polar and non-polar. PF5 has a trigonal bipyramidal configuration consisting of a central phosphorus atom surrounded by five fluorine atoms. PF5 is a nonpolar molecule because there is no net dipole moment in the molecule.
Agray339 agray339 12082018 Chemistry Middle School answered What is the geometry name for PF5. Add your answer and earn points. PF5 is a nonpolar molecule because when you consider its horizontal and vertical planes electron pulls balance themselves out to achieve a neutral conformation.
For each polar molecule choose the molecule and Common Cases of Adding Dipole Moments to Determine Non-Polar Molecule. By the reaction of FSO3H on fluoride and phosphate. Polar In chemistry polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment.
Is PF5 polar or non-polar. Phosphorus pentafluoride was first prepared in 1876 through fluorination of phosphorus pentachloride using arsenic trifluorideOther routes to PF5 have included fluorination of PCl5 by HF AgF benzoyl fluoride SbF3 PbF2 or CaF2It can also be made by the reaction of PF3 and fluorine chlorine or chlorine in contact with calcium fluoride. As the shape of the molecule is symmetric with even charge distribution it makes Phosphorus Pentafluoride a nonpolar molecule.
Pf5 is not polar. The electronegativities of the five fluorine-phosphorus bonds cancel each other out creating a nonpolar molecule. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.
Get the answers you need now. Hey Guys In this video we are going to determine the polarity of Phosphorus Pentafluoride having a chemical formula of PF5To know the polarity of this mole. As a result all the P-F bonds ensure non zero dipole moment in the same direction.
Due to the lone pair present on phosphorus atom the bonds face a repulsive force in a downward direction due to repulsion between lone pair and bond pairs. Due to this difference between their electronegativity the P-F bond is polar. Question Is PF5 polar or nonpolar.
Phosphorus pentafluoride which is abbreviated as PF5 is a nonpolar molecule. Imagines it to have the same geometry as PF5 namely trigonal bipyramidal then by symmetry PH5. 1 See answer agray339 is waiting for your help.
Let me explain why. Answer IF5 Iodine pentafluoride is Polar What is polar and non-polar. PH5 doesnt actually exist as a stable molecule.
The Phosphorus Pentafluoride PF5 is a nonpolar molecule because the bond P-F is polar and the F atom in PF5 has more electronegative than P. Is PF5 polar or non-polar. Is PH5 a polar molecule.
When a molecule have a original molecular geometry like linear sp trigonal planar sp2 tetrahedral trigonal bi-pyramidal and octahedral with the same type of ligands in your case flourine they will be non polar. However the fluorine atoms in PF5 spread out to form a trigonal bipyramidal shape because there are no lone pairs on the phosphorous. Answer C2Cl4 Tetrachloroethylene is nonPolar What is polar and non-polar.
Altherra Altherra Geometry name is trigonal bypyramidal non-polar bipyramidal. If a molecule has four hybrid sp3 orbitals it can For each polar molecule choose the molecule and The molecular geometry of the CHF3 molecule is The unequal sharing of electrons within a water. Therefore PF5 is a polar molecule.
Polar In chemistry polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment.

Frozen Silver F80 M3 2016 Frozen Silver W07 7 Bmw M3 Sedan Bmw 328i Bmw Cars

Is Co Polar Or Nonpolar Carbon Monoxide In 2021 Carbon Carbon Monoxide Polar

Is Sf4 Polar Or Non Polar Sulfur Tetrafluoride In 2021 Math Equations Chemical Formula Molecules

Hybridization Of Ch3cl Chloromethane In 2021 Molecules Lewis Chemical Formula

Frozen Silver F80 M3 2016 Frozen Silver W07 7 Bmw M3 Sedan Bmw 328i Bmw Cars

Cs2 Lewis Structure Carbon Disulfide In 2021 Lewis Math Equations Molecules

P4 Lewis Structure Tetraphosphorus In 2021 Molecules Lewis Electrons

Armored Car Humvee Guntruck Iraq Armored Vehicles Work Truck Military Vehicles

Is Ch3oh Polar Or Nonpolar Methanol In 2021 Functional Group Molecules Chemical Formula

Ch2o Lewis Structure Methanal Or Formaldehyde In 2021 Methanal Molecules Lewis

Is Pf5 Polar Or Non Polar Phosphorus Pentafluoride In 2021 Chemical Formula Molecules Phosphorus
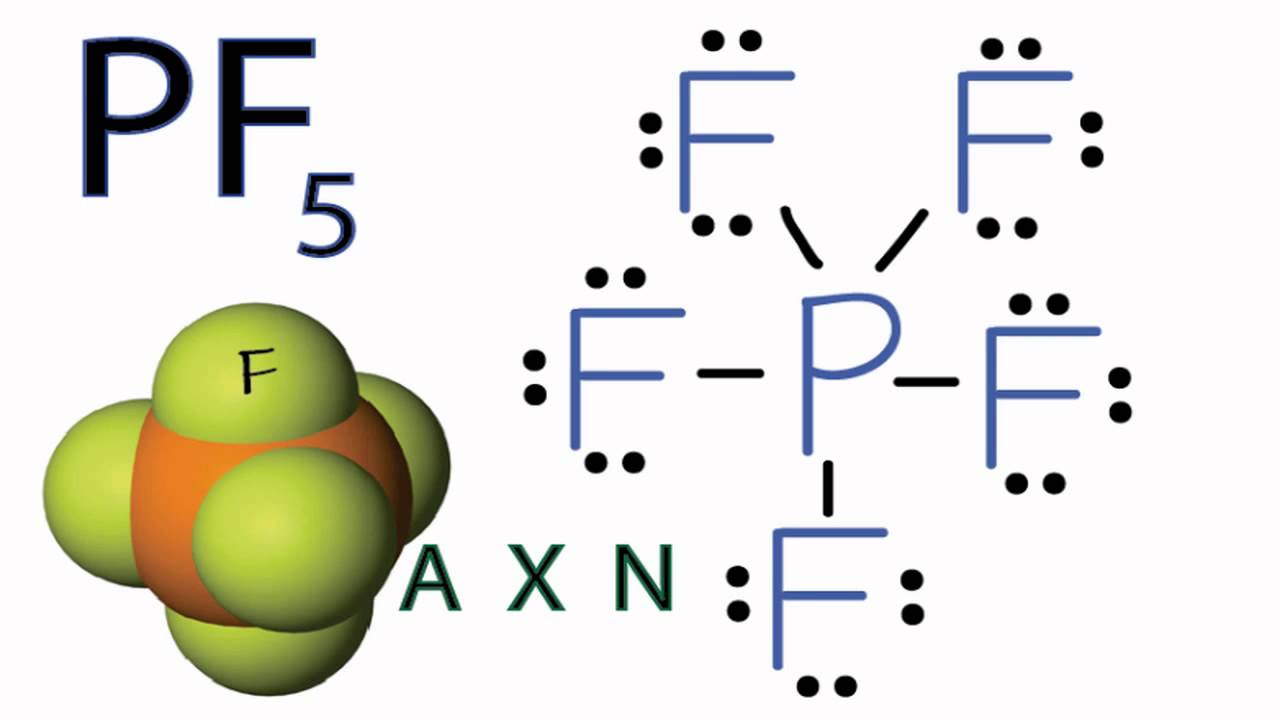
Pf5 Molecular Geometry Shape And Bond Angles Molecular Geometry Geometry Shape Molecular